In what could eventually become a milestone for H.I.V. prevention, very preliminary tests of an implant containing a new drug suggest that it may protect against infection for a full year.
The new implant, by the drug company Merck, was tested in just a dozen subjects for 12 weeks. But experts were quite excited at its potential to revolutionize the long battle against H.I.V.
The research was described on Tuesday at an international AIDS conference in Mexico City.
New H.I.V. prevention methods are desperately needed. About 75 million people have contracted the lethal virus since the AIDS epidemic began. Even now, about 1.7 million people are infected each year — despite decades of promotion of condoms and abstinence, and years of efforts to get people to take a daily pill that prevents infection.
“If — and I’m emphasizing if — if it pans out in a larger trial that it delivers a level of drug that’s protective for a year, that would be a game-changer,” said Dr. Anthony S. Fauci, director of the National Institute for Allergy and Infectious Disease and a leading expert on AIDS.
Dr. Robert M. Grant, a researcher at the University of California, San Francisco, who led the landmark 2010 trial that proved that a daily pill could thwart H.I.V., said he had expected a breakthrough based on the powerful new drug used in the implant.
The device “seems ideal in many respects,” he added. “It can be removed if there are side effects or H.I.V. infection.”
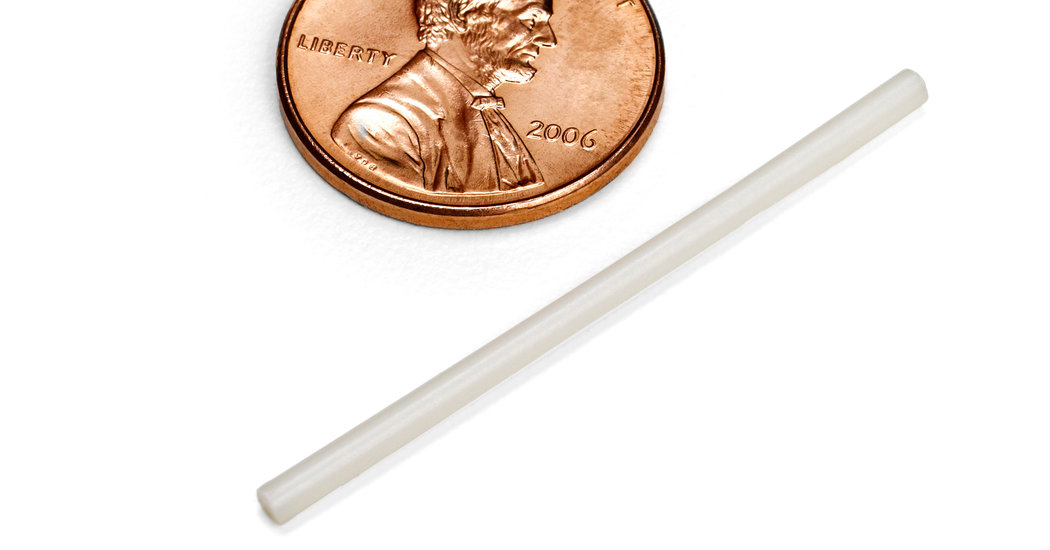
Merck’s innovation is to deliver its new antiretroviral drug, islatravir, with a proven technology long used for birth control: a matchstick-sized plastic rod inserted just under the skin of the upper arm that slowly releases tiny doses of the medication.
Many people at risk of H.I.V. infection, particularly women in Africa, are desperate for prevention methods that are easier to use, and easier to conceal, than a bottle of pills.
According to Unaids, more than 6,000 young women under age 24 are infected every week, and 80 percent of infected teenagers in Africa are girls.
African girls are often victims of rape or pressured into sex with older men in return for food, clothes or cash.
Another study released Tuesday at the same conference reinforced the extreme vulnerability of young African women.
In it, 427 women and teenage girls in South Africa and Zimbabwe were given Truvada pills, the form of so-called pre-exposure prophylaxis to prevent H.I.V. infection, also called PrEP, used in the United States.
They were repeatedly reminded to take the pills every day, but after one year, blood tests showed that less than a third were still taking any pills. Only 9 percent of the women and girls took them frequently enough to have protective levels of the drug in their bloodstreams.
The study’s authors are still interviewing participants about why they failed to comply. But women in Africa often say they cannot keep H.I.V. drugs at home because they fear being accused by family, neighbors and lovers of being immoral or of having H.I.V. Some fear being threatened or beaten.
(Clinical trials among women in Africa often fail, or produce hard-to-interpret results, because some participants sign up but rarely use the interventions being tested. In interviews afterward, some have admitted to joining only because the trials offer free medical care and modest payments for participating.)
The drug used in the new implants is islatravir (pronounced IZ-lah-trah-veer), which until last week was known as EFdA or MK-8591. It is the first in a new class of drugs called nucleoside reverse transcriptase translocation inhibitors, which block movement of the enzyme responsible for cloning the virus’s DNA so that it can infect new cells.
Islatravir has “some remarkable attributes,” said Dr. Roy D. Baynes, Merck’s chief medical officer.
It is 10 times as potent as any previous H.I.V. drug, he said, so tiny amounts are effective, which lowers the risk of side effects. The drug lingers in the body for a relatively long time — after five days, half of the dose remains — so it can be given less often that other H.I.V. medications.
Unlike some H.I.V. drugs, islatravir is absorbed into anal and genital tissues, which is where most infections start. And because it attacks a different step in the infection process, the drug appears not to give rise to viral strains with cross-resistance to other H.I.V. drugs.
Other drug companies have recently reported success with other long-acting forms of H.I.V. prevention. For example, injections of cabotegravir or rilpivirine deep into the buttocks have protected study participants for a month.
But monthly intramuscular injections are inconvenient and sometimes painful. And implants can be removed. The half-life of injectable cabotegravir, for example, is about 40 days. If a user develops a “breakthrough” H.I.V. infection in spite of the injection, the lingering drug may drive the virus to mutate into a resistant form.
Although scientists are excited about the Merck implant’s possibilities, they warned that it still needs to prove itself.
The drug has protected rats and monkeys against multiple deliberate attempts to infect them, but human testing thus far has been only for safety: to see whether a handful of people could tolerate the implant for three months without dangerous side effects.
The assumption that the implant could protect against H.I.V. for a year was arrived at by estimating how much drug is considered protective and how much the implant can hold. A real-world test will mean giving the implant to thousands of sexually active or drug-using men and women, and tracking how many get infected.
Such a trial will be long and expensive, said Mitchell J. Warren, executive director of AVAC, an advocacy group for H.I.V. prevention.
Participants cannot just be given the implant and told to go out and have sex or inject drugs. Ethically, they must be offered the best prevention methods available, including oral PrEP, condoms, clean syringes and intensive counseling about safe sex or safe drug use.
That means many subjects must be followed for years to see if the implant works better than other methods.
[Like the Science Times page on Facebook. | Sign up for the Science Times newsletter.]
Dr. Baynes, of Merck, declined to discuss how the next trial would be designed or to speculate on how long it would take. The company, he said, would consider testing in both the United States, where most H.I.V. transmission is between gay men, and in Africa, where the virus is passed mostly between men and women.
He also declined to discuss the eventual cost, other than to say “we’re committed to responsible pricing.”
Price is a deeply contentious issue in PrEP. High hopes for a quick end to the AIDS epidemic raised by Dr. Grant’s 2010 study were dashed after relatively few Americans began taking Truvada — in part because Gilead, the patent-holder, raised the price to $ 20,000 a year and sued every generic competitor that tried to enter the market.
Other research groups have begun testing similar subdermal implants for H.I.V. prevention, such as one that contains a new form of tenofovir, the main drug in Truvada.
High prices for prevention methods have been a constant disappointment, Dr. Grant said: “There is always a bright shiny new object with people asking, ‘Who cares what it costs?’”
